
The Future of Signal Management: How Process Automation is Shaping the Pharmacovigilance Landscape
Signal management is a fundamental aspect of pharmacovigilance, helping pharmaceutical
Despite advances in automated detection technology, safety teams continue to use outdated and manual tools for the rest of their workload. This puts them at risk of missed deadlines, reduced quality and overlooked data.
Orbit takes a different approach to signal management:
Orbit works in parallel with your detection system to add automation, workflow support and compliance to your end-to-end process.

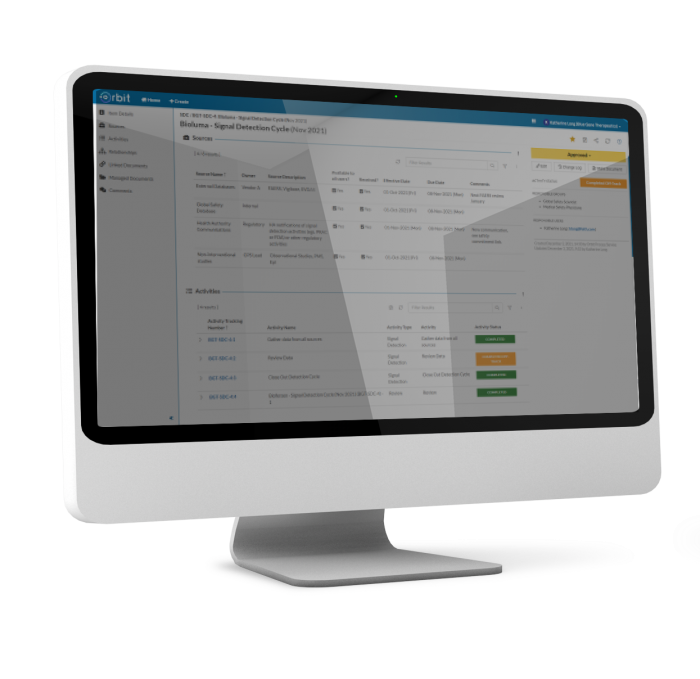
Manage the flow of all activities and data around detected signals, including analysis, prioritization and validation. Orbit helps you and your team track potential and validated signals from detection through resolution and closure.
Orbit helps you define your signal detection strategy for each product. Make key signaling stakeholders, partners and data sources available at-a-glance. One location stores all source information, helping your team execute your Safety Surveillance Strategy on time and with a full, reportable, audit trail.
01
Create product detection strategy
02
Execute recurring surveillance activities
03
Track evidence, outcomes, and decisions
Reliable access to your decisions, data, and compliance obligations.
Manage your signal detection process across platforms, tools, and stakeholders
Store evidence and meeting minutes from detection, validation and assessment centrally
Add automation to your SOP's to deliver high quality decisions and process efficiency

Signal management is a fundamental aspect of pharmacovigilance, helping pharmaceutical

The pharmacovigilance industry is locked in a race toward ever-more

Most Pharma companies rely on passive tools—such as spreadsheets and

WEBINAR Enabling a modernized signal management process with Orbit On

WEBINAR The Digital Transformation Toolkit for Global Patient Safety On-demand

WEBINAR Signal Management with Orbit Safety Signals are the driving
Notify local partners and affiliates of newly detected or validated safety signals.

Track Safety Commitments and Health Authority Requests across Regulatory and PV functions.

With integrated MedDRA browsing, manage lists of structured query strategis.
Schedule a call today.
Once we’ve received your registration, login information will be sent to your inbox.