
The Hidden Risks of Noncompliance: Why Insight into Pharmacovigilance Agreement Responsibilities Matters
In the pharmaceutical industry, ensuring patient safety is non-negotiable. Pharmacovigilance
For companies with many partners and vendors, managing the workload around pharmacovigilance agreements is complex. With many data points, commitments, and deadlines spread across hundreds of global contracts, maintaining and demonstrating compliance is a constant challenge.
Orbit streamlines PVA oversight with a centralized database of contracts, responsibilities, and compliance status.
01
Manage PVA versions and approvals
02
Implement negotiated activities
03
Oversee global status and compliance


No need to search through contracts to determine your delegated responsibilities: Orbit stores this in a structured database to give you instant access to your information. Quickly access responsibilities across markets, products, and partners.
Monitor PVA health for all partners across your global organization. With recurring reminders, automated notifications and streamlined data collection, Orbit automates manual work and helps demonstrate PVA compliance.
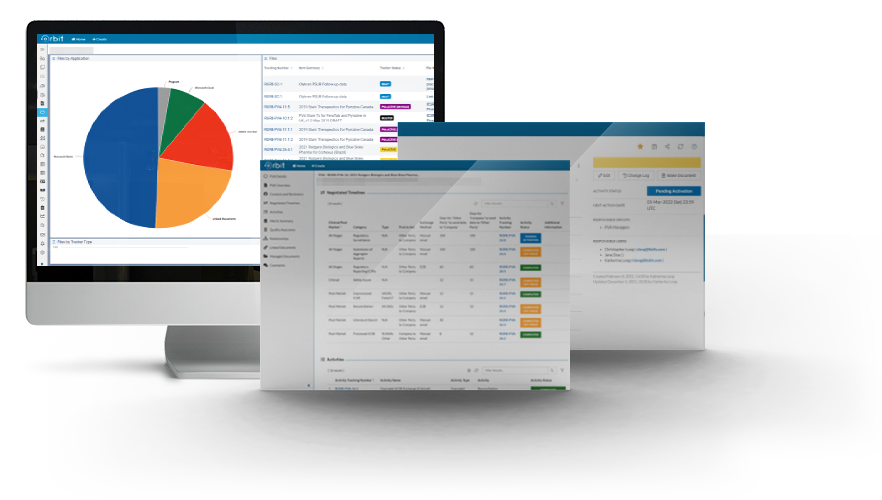
See the big picture of your PVAs in a glance with dashboards and reports. Orbit ‘s data visualizations provide your team with oversight of global PVA Status, compliance and upcoming version reviews.
Orbit helps guide your team from drafting, negotiation, and approval of PV Agreements through one simple interface. Manage agreements with proactive reminders, version comparison, and retirement workflows.
Authoring and review workflows
Versioning and update management
Partnership retirement workflows

In the pharmaceutical industry, ensuring patient safety is non-negotiable. Pharmacovigilance

Pharmacovigilance Agreements (PVAs) are essential tools in ensuring drug safety

Unlocking the complexity of Pharmacovigilance Agreements through five essential phases.

WEBINAR PVA Mastery for Oversight: Streamlining Agreements, Tracking, and Reporting

WEBINAR Revolutionize Your Business Partner Agreement Process On-Demand Are you

WEBINAR PVAs: Looking Beyond a CAPA with Zina Sadeq of
Schedule a call today.
Once we’ve received your registration, login information will be sent to your inbox.